Regulatory Strategy & Submissions
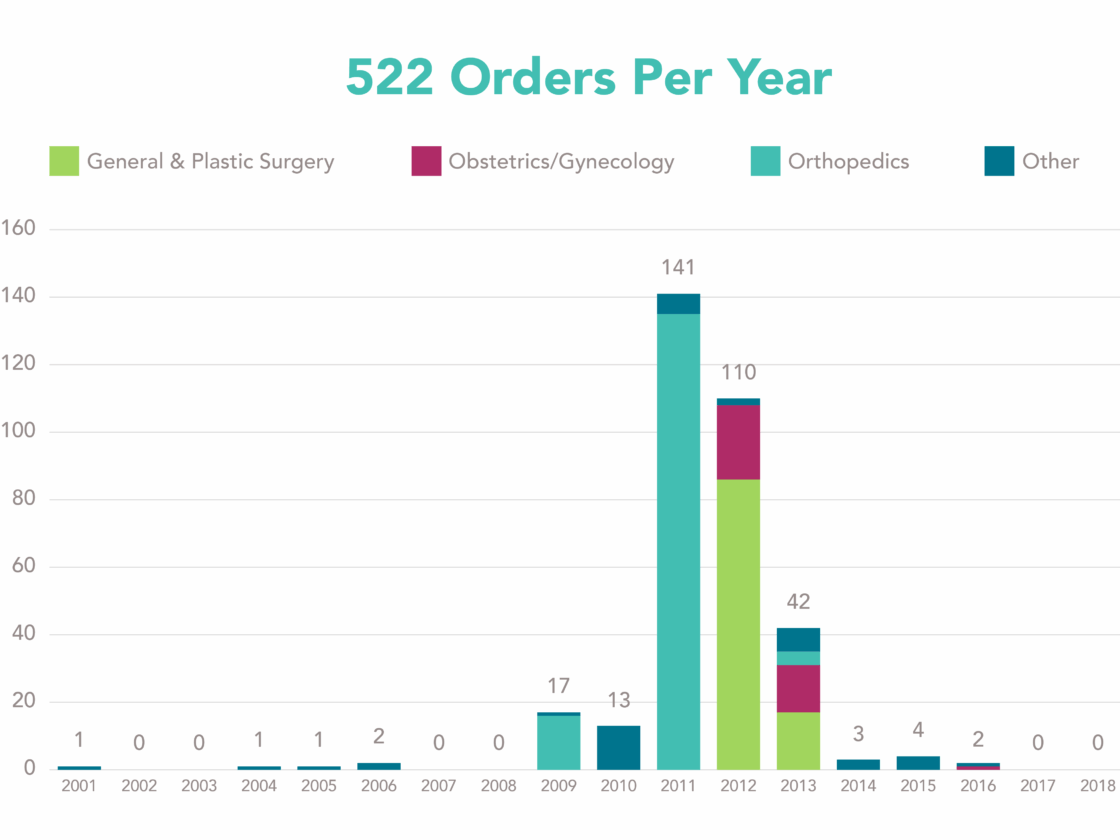
Post-market surveillance under a “522 order” — why, what, and how?
Company X has gained 510(k) clearance for their new heart valve and was notified by the Food and Drug Administration...
Medical apps under the new European MDR
Worldwide, one out of every three internet users currently monitors their health or fitness using an app. This was shown in...
Clinical Evaluation Plan – The Start of All Clinical Evaluations
As manufacturer or developer of a medical device, you may already have experienced that providing enough clinical evidence that your...
Have You Considered an EUA for Your Diagnostic Test?
In certain emergencies, like the current COVID-19 pandemic, the FDA can quickly issue an Emergency Use Authorization for diagnostic tests...
Is the FDA’s EFS Program Right for Your Medical Device?
What is an Early Feasibility Study (EFS)? The FDA created its EFS program to help increase patient access to new,...
Global Regulatory Update: A Trend towards Harmonization and Enforcement
Gaining access to the global medical device market can be challenging because regulatory requirements are rapidly changing. Medical device manufacturers must be well acquainted with…
Environmental and Social Considerations for Medical Devices
Get Your Copy Now Name(Required) First Name Last Name Company name(Required)Email(Required) Consent(Required) By checking here, you agree for the information...
Final FDA Guidance – Post-Market Surveillance Under Section 522 of the Federal Food, Drug, and Cosmetic Act
Get Your Copy Now Name(Required) First Name Last Name Company name(Required)Email(Required) Consent(Required) By checking here, you agree for the information...
Changes to Medical Device Regulations in China
Get Your Copy Now Name(Required) First Name Last Name Company name(Required)Email(Required) Consent(Required) By checking here, you agree for the information...
Factors to Consider When Making Benefit-Risk Determinations in Medical Device Premarket Approval and De Novo Classifications
The FDA confirms that the potential benefit of a medical device outweighs the potential risk before they will approve the device. The FDA considers several…